
Top ▲

GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Gene and Protein Information  |
|||||||
| Species | TM | P Loops | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | 3 | 1 | 908 | 6q16.3 | GRIK2 | glutamate ionotropic receptor kainate type subunit 2 | |
| Mouse | 3 | 1 | 908 | 10 24.87 cM | Grik2 | glutamate receptor, ionotropic, kainate 2 (beta 2) | |
| Rat | 3 | 1 | 908 | 20q13 | Grik2 | glutamate ionotropic receptor kainate type subunit 2 | |
Database Links  |
|
| Alphafold | Q13002 (Hs), P39087 (Mm), P42260 (Rn) |
| ChEMBL Target | CHEMBL3683 (Hs), CHEMBL3607 (Rn) |
| Ensembl Gene | ENSG00000164418 (Hs), ENSMUSG00000056073 (Mm), ENSRNOG00000000368 (Rn) |
| Entrez Gene | 2898 (Hs), 14806 (Mm), 54257 (Rn) |
| Human Protein Atlas | ENSG00000164418 (Hs) |
| KEGG Gene | hsa:2898 (Hs), mmu:14806 (Mm), rno:54257 (Rn) |
| OMIM | 138244 (Hs) |
| Orphanet | ORPHA205590 (Hs) |
| Pharos | Q13002 (Hs) |
| RefSeq Nucleotide | NM_021956 (Hs), NM_010349 (Mm), NM_019309 (Rn) |
| RefSeq Protein | NP_068775 (Hs), NP_034479 (Mm), NP_062182 (Rn) |
| SynPHARM | 83004 (in complex with kainate) |
| UniProtKB | Q13002 (Hs), P39087 (Mm), P42260 (Rn) |
| Wikipedia | GRIK2 (Hs) |
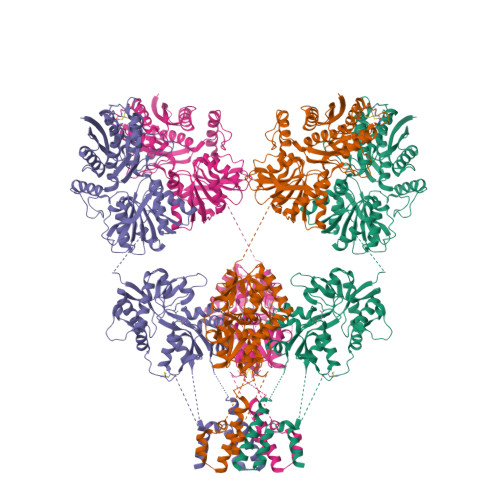
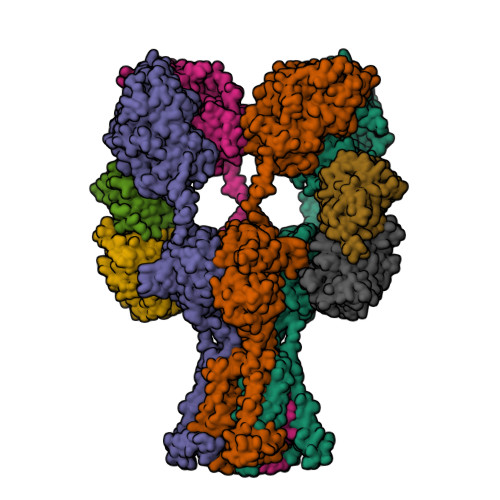
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||

|
|
||||||||||||
|
|
||||||||||||
Natural/Endogenous Ligands  |
| L-glutamic acid |
Download all structure-activity data for this target as a CSV file 
| Agonists | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| View species-specific agonist tables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonists | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Allosteric Modulators | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||
|
||||||||||||
1. Cantrell BE, Zimmerman DM, Monn JA, Kamboj RK, Hoo KH, Tizzano JP, Pullar IA, Farrell LN, Bleakman D. (1996) Synthesis of a series of aryl kainic acid analogs and evaluation in cells stably expressing the kainate receptor humGluR6. J Med Chem, 39 (19): 3617-24. [PMID:8809152]
2. Gangwar SP, Yelshanskaya MV, Nadezhdin KD, Yen LY, Newton TP, Aktolun M, Kurnikova MG, Sobolevsky AI. (2024) Kainate receptor channel opening and gating mechanism. Nature, 630 (8017): 762-768. [PMID:38778115]
3. Gangwar SP, Yen LY, Yelshanskaya MV, Sobolevsky AI. (2023) Positive and negative allosteric modulation of GluK2 kainate receptors by BPAM344 and antiepileptic perampanel. Cell Rep, 42 (2): 112124. [PMID:36857176]
4. Larsen AP, Fièvre S, Frydenvang K, Francotte P, Pirotte B, Kastrup JS, Mulle C. (2017) Identification and Structure-Function Study of Positive Allosteric Modulators of Kainate Receptors. Mol Pharmacol, 91 (6): 576-585. [PMID:28360094]
5. Meyerson JR, Kumar J, Chittori S, Rao P, Pierson J, Bartesaghi A, Mayer ML, Subramaniam S. (2014) Structural mechanism of glutamate receptor activation and desensitization. Nature, 514 (7522): 328-34. [PMID:25119039]
6. Pedregal C, Collado I, Escribano A, Ezquerra J, Domínguez C, Mateo AI, Rubio A, Baker SR, Goldsworthy J, Kamboj RK et al.. (2000) 4-Alkyl- and 4-cinnamylglutamic acid analogues are potent GluR5 kainate receptor agonists. J Med Chem, 43 (10): 1958-68. [PMID:10821708]
7. Sagot E, Pickering DS, Pu X, Umberti M, Stensbøl TB, Nielsen B, Chapelet M, Bolte J, Gefflaut T, Bunch L. (2008) Chemo-enzymatic synthesis of a series of 2,4-syn-functionalized (S)-glutamate analogues: new insight into the structure-activity relation of ionotropic glutamate receptor subtypes 5, 6, and 7. J Med Chem, 51 (14): 4093-103. [PMID:18578478]
8. Sakai R, Swanson GT, Shimamoto K, Green T, Contractor A, Ghetti A, Tamura-Horikawa Y, Oiwa C, Kamiya H. (2001) Pharmacological properties of the potent epileptogenic amino acid dysiherbaine, a novel glutamate receptor agonist isolated from the marine sponge Dysidea herbacea. J Pharmacol Exp Ther, 296 (2): 650-8. [PMID:11160654]
9. Small B, Thomas J, Kemp M, Hoo K, Ballyk B, Deverill M, Ogden AM, Rubio A, Pedregal C, Bleakman D. (1998) LY339434, a GluR5 kainate receptor agonist. Neuropharmacology, 37 (10-11): 1261-7. [PMID:9849663]
10. Zhou LM, Gu ZQ, Costa AM, Yamada KA, Mansson PE, Giordano T, Skolnick P, Jones KA. (1997) (2S,4R)-4-methylglutamic acid (SYM 2081): a selective, high-affinity ligand for kainate receptors. J Pharmacol Exp Ther, 280 (1): 422-7. [PMID:8996224]