GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Transient Receptor Potential channels (TRP): Introduction
INTRODUCTION
The superfamily of transient receptor potential (TRP) ion channels consists of 28 distinct genes in mammals. They are grouped into six subfamilies by sequence homology: Canonical (TRPCs), Vanilloid (TRPVs), Melastatin (TRPMs), Mucolipins (TRPMLs), Polycystins (TRPPs), and Ankyrin repeat (TRPA). TRP channels may be generally described as cation permeable channels that function as cellular sensors responding to a broad range of stimuli.
Transient receptor potential (TRP) channels constitute a superfamily of cation permeable ion channels. They integrate multiple stimuli as cellular sensors, are widely expressed, and function in many physiological processes [15,24]. While a subset of TRP channels primarily spans organellar membranes such as endolysosomes, most TRP channels promote Na+ and Ca2+ flux into cells [56]. In this article, we summarize molecular, structural, and functional properties of TRP ion channels.
DISCOVERY AND CLASSIFICATION
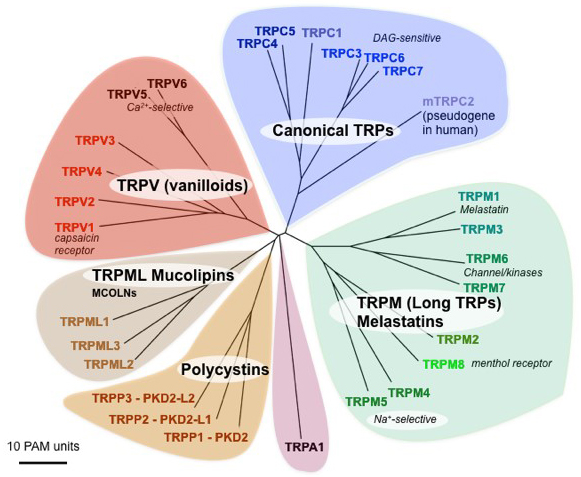
The founding member of the TRP channel superfamily was identified as essential component of Drosophila phototransduction [40]. A spontaneous mutation in the trp gene resulted in a transient receptor potential in response to continuous light. Drosophila TRP homologs have been identified in yeast, invertebrates and vertebrates. In mammals, 28 genes are classified as TRP channel subunits that are grouped into six subfamilies: the canonical TRPs (TRPCs), the vanilloid receptor TRPs (TRPVs), the melastatin TRPs (TRPMs), the mucolipins (TRPMLs), the polycystins (TRPPs), and the ankyrin transmembrane protein 1 (TRPA1) [15] (Figure 1). Unlike other ion channels that are classified according to a common ligand, function, or selectivity, TRP channel classification is currently based only on sequence homology [17,80]. TRPCs, TRPVs, TRPMs, and TRPA1 have the highest homology to Drosophila TRP with TRPCs being the closest homologs. TRPMLs and TRPPs are more distantly related [75].
Figure 1. Mammalian TRP ion channel family tree. The branch lengths in point accepted mutations (PAM, the mean number of substitutions per 100 residues) shows the evolutionary distance. DAG, diacylglycerol. Modified from Clapham DE (2003) TRP channels as cellular sensors. Nature 426: 517-524.
The TRPC subfamily comprises seven members and can be further divided into 3 subgroups based on homology and functional similarities: TRPC1/C4/C5, TRPC3/C6/C7, and TRPC2 (pseudogene in humans). TRPC1 forms heteromeric channels with TRPC4 and TRPC5, but whether it functions as homomer is unclear [66-67]. The TRPV subfamily has six members that can broadly be divided into the non-selective cation channels, TRPV1-V4 and the more Ca2+-selective channels TRPV5 and TRPV6. The TRPM subfamily contains eight members that may be grouped into 4 subgroups: TRPM1/M3, TRPM2, TRPM4/M5, TRPM6/M7, and TRPM8. TRPM1 appears to form an intracellular channel [47]. TRPM6 and TRPM7 are unique among TRPs, and indeed among ion channels, since they are 'chanzymes' comprised of a channel fused to a kinase domain [41,58]. The TRPML subfamily has three members, TRPML1-ML3 and are mostly confined to intracellular (endo-lysosomal) compartments [82]. The TRPP subfamily comprises three members, TRPP1, TRPP2, and TRPP3 that are also known as PKD2, PKD2-L1, and PKD2-L2 (TRPP2, TRPP3, TRPP5 according to the old nomenclature; [80]). Originally, TRPPs included the PKD1 (polycystic kidney disease 1, polycystin 1; PC1) family since those molecules contained a putative channel-like region. In the absence of any proven channel function, however, we considered it prudent to only include the PKD2 (PC2, polycystin 2) subfamily. TRPA1 is the sole member of the TRPA subfamily in mammals.
STRUCTURAL FEATURES
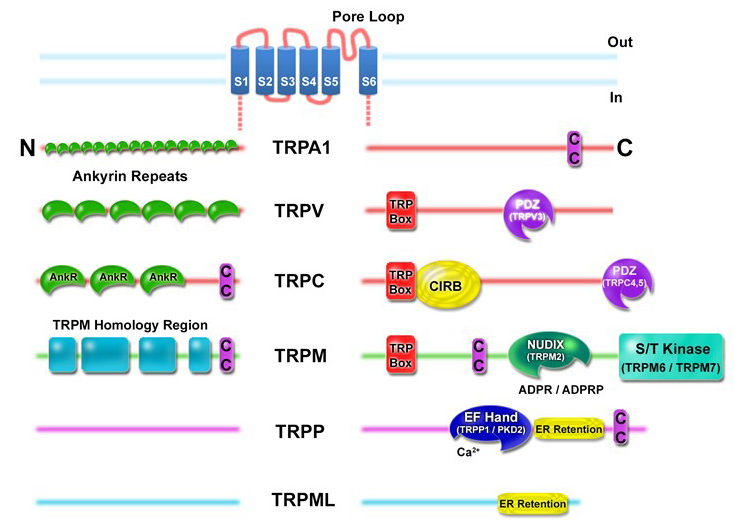
All TRP channel subunits are predicted to contain 6 transmembrane spanning domains (S1-S6) (Figure 2). The pore region is formed by a stretch of hydrophobic residues between S5 and S6. With the exception of the Na+-selective TRPM4 and TRPM5, and the Ca2+-selective TRPV5 and TRPV6, all TRPs are non-selective cation channels [15]. The cytoplasmic termini of TRP subunits contain different structural and functional domains that vary between subfamilies [15]. A 25-amino acid motif, defined as TRP domain, located C-terminal to the sixth transmembrane segment is loosely conserved in almost all TRP channels, except TRPA1 and the distantly related TRPMLs and TRPPs [56]. A stretch of 6-amino acids within the TRP domain defined as TRP box is highly conserved among TRPCs (EWKFAR), but less conserved in TRPMs and TRPVs. The primary structure of TRPA1 lacks the canonical TRP box motif, but recent cryo-EM analysis revealed a domain adjacent to S6 that is topologically similar to the TRP domain in TRPV1 [49]. Another feature shared by TRPCs, TRPVs, and TRPA1 are N-terminal ankyrin repeat domains. Ankyrin repeats which generally function as a scaffold for protein-protein interactions may contribute to TRP channel regulation and subunit assembly [6-7,21,33,49]. Other structural features include coiled-coil domains, calmodulin and Ca2+ binding motifs, C-terminal PDZ-binding domains (TRPC4/C5), serine/threonine kinases (TRPM6/M7), and a Nudix hydrolase domain (TRPM2) [15] (Figure 2). While ankyrin repeats (except TRPM) and TRP domains are common to TRP subfamilies (TRPCs, TRPVs, TRPMs, and TRPA1) that are more closely related to Drosophila TRP, a large extracellular loop between S1 and S2 is a common feature of the two more distantly related TRP subfamilies, TRPMLs and TRPPs [75].
Figure 2. TRP channel domains. TRP channels have a predicted 6 transmembrane topology (S1-S6) with a pore loop between S5 and S6. The cytoplasmic N- and C-termini contain different structural and functional elements as indicated for each subfamily. TRP Box, EWKFAR in TRPCs; AnkR, ankyrin repeats (number differs by subfamily members); CC, coiled-coil domain (position in the cytoplasmic termini varies); S/T Kinase, serine/threonine kinase intrinsic to TRPM6 and TRPM7; CIRB, calmodulin- and inositol triphosphate receptor (InsP3R)-binding site; PDZ, amino acid motif binding PDZ domains (TRPV3 contains a consensus PDZ binding motif); NUDIX, NUDT9 homology domain binding ADP ribose (ADPR) or ADPR-2'-phosphate (ADPRP); EF Hand, canonical Ca2+-binding domain in TRPP1/PKD2 (EF Hand sequence motif in TRPA1 not shown); ER retention, endoplasmic reticulum retention signal. The TRPM Homology Region is of unknown function. Modified from Clapham DE (2003) TRP channels as cellular sensors. Nature 426: 517-524.
Three-dimensional structure
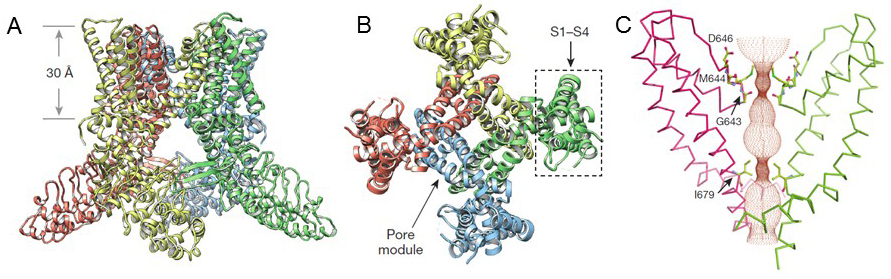
Recent cryo-EM structures for TRPV1, TRPV2, and TRPA1 [7,26,33,49,85] at near atomic resolution illustrate that TRP channels adopt a similar architecture to voltage-gated K+ ion channels [35-36]. Four subunits assemble as a tetramer around a central pore, which is formed by S5, a pore helix, and S6, and flanked by the S1-S4 helices (Figure 3). Based on the structures for TRPV1, TRPV2, and TRPA1, the TRP channel pore has two constrictions with the upper gate being formed by the selectivity filter in the outer pore region and the lower gate by the distal end of S6 [7,26,33,49,85] (Figure 3).
The pore profiles of TRPV1 in distinct states support a dual gating mechanism with conformational changes at the upper and lower gate [7,33]. Opening of the upper gate is associated with movements in the outer pore and pore helix, while movement of the S4-S5 linker and concomitant conformational changes of the lower half of the S6 helix are presumed to result in expansion of the lower gate, which is accompanied by a lateral displacement of the TRP domain [7,33]. Unlike voltage-gated ion channels, in which opening and closing of the gate is coupled to S1-S4 voltage sensor movement [11], the S1-S4 domain may be static in TRPV1 [7,33], consistent with a lack of classic voltage-sensing elements in S4 and weak voltage-dependence of TRPV1 [7,44]. In general, TRP channel gating is not dominated by voltage, but rather effected by the coupling of their voltage sensitivity to the energy differences accompanying changes in temperature or ligand binding which can shift their gating towards a physiologically relevant range [44,80].
The most unique features of the TRP channels are their cytoplasmic domains. The conserved C-terminal TRP domain forms an α-helical structure that runs perpendicular to the pore helices and may contribute to regulation of the lower gate [7,16,33,49]. The coiled-coil domain in the C-terminal region of TRPA1 mediates bundling of the four subunits and is surrounded by at least 16 ankyrin repeat domains. The ankyrin repeats in TRPA1 may communicate with the pore through interactions with the helix-turn-helix motifs of the linker region that propagate into a web of interactions with the TRP domain and overlying S4-S5 linker [49]. In TRPV1, one of the ankyrin repeats may contribute to subunit interaction through contact with a three-stranded β-sheet on an adjacent subunit [7,33].
Figure 3. TRPV1 structure. A, B) Ribbon diagrams showing the TRPV1 structure in the apo (closed) state in side view (A) or bottom view (B). Subunits in A and B are color-coded. C) TRPV1 pore with marked ion permeation pathway. The two sites of constriction highlighting the upper and lower gate are indicated. From Liao et al. (2013) Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504:107-112, with permission.
ACTIVATION MECHANISMS AND PHARMACOLOGY
Most TRP channels can be activated and/or modulated by multiple stimuli including receptor coupled intracellular signaling pathways, physical, and chemical stimuli.
It is generally accepted that all TRPCs are activated downstream of G protein-coupled receptors (Gq-linked) and receptor tyrosine kinases, but other TRPs have also been shown to be modulated by phospholipases C (PLC) mediated hydrolysis of phosphatidylinositol 4,5-bisphosphate (PI(4,5)P2), and production of diacylglycerol (DAG) and inositol-1,4,5-triphosphate (IP3) [56]. PI(4,5)P2 is a common regulator of many ion channels and not specific to TRP channels. The reported effects of PI(4,5)P2 on TRP channels are often complex, occasionally contradictory, and likely to be dependent upon experimental conditions, such as intracellular ATP levels (reviewed by [43,57]).
TRPC channels have frequently been proposed to act as store-operated channels (SOCs) (or components of multimeric complexes that form SOCs), activated by depletion of intracellular calcium stores (reviewed by [1,12,50,55,59,83]). However, the weight of the evidence is that they are not directly gated by conventional store-operated mechanisms, as is established for Stim-gated Orai channels (also known as CRAC channels).
Among the physical stimuli that activate TRP channels, temperature is one of the most remarkable stimuli. Several TRP channels (TRPV1-V4, TRPC5, TRPM3, TRPM5, TRPM8, TRPA1) [9-10,25,28,38,51-52,63-64,70,76,78,81,84] exhibit temperature sensitivity with high Q10s of gating (reviewed by [23]). Defined as the change in the rate of a reaction when temperature is changed by 10°C, the Q10 value provides an estimate for the temperature sensitivity of a protein. While the thermal activation thresholds in thermosensitive TRP channels cover the range of temperatures sensed (from cold to hot), the high Q10s of gating do not necessarily imply a function as a 'hot' or 'cold' sensor. Protein folding is of course temperature sensitive, and thus a change in gating within the narrow temperature range experienced by organisms does not dictate that they are sensors for temperature. For example, many channels with high Q10s are present in the mammalian CNS and other isothermal regions where they are not exposed to temperatures outside a narrow range around 37°C (see [18] for discussion).
Mechanosensitivity has also been proposed for several mammalian TRP channels (detailed in [54]). Like temperature, all proteins are sensitive to mechanical forces, but these are only relevant if these forces are in the range experienced by proteins in their native membranes and locations. Those often cited as mechanosensitive include TRPP1 (PKD2), TRPA1, TRPV4, and TRPV1, but relevant experimental evidence for physiological significance is often lacking or controversial. In contrast, MscL, MscS (bacterial), Piezo1 and 2, and hair cell channels (putatively TMC1/2) are clearly important to physiologically relevant mechanosensing [19-20,29,31,68-69].
Endogenous and exogenous molecules activate TRP channels. Although progress has been made in uncovering endogenous ligands and signaling pathways that trigger TRP channel activation, it is often still unclear what gates the channels in vivo [80]. Among the known endogenous activators or modulators are inorganic ions (mainly Ca2+ and Mg2+), bioactive lipids (eicosanoids, diacylglycerol, anandamide), and ADP ribose or ADP ribose-2'-phosphate [56,73]. Increases in intracellular Ca2+ can activate some TRP channels, but will modulate, directly or via calmodulin, almost all TRP channels [80].
Several natural products (e.g. capsaicin, menthol, or carvacrol), but also synthesized small molecules have been shown to affect TRP channel gating. However, a selective pharmacology does not yet exist for most TRP channels. Commonly used blockers such as Ruthenium red or 2-APB (2-Aminoethoxydiphenyl borate) are highly nonspecific. More useful tools with high potency and more selectivity have just started to emerge. Examples are the TRPV4 agonist GSK1016790A or its antagonists GSK2193874 and HC-067047, and the TRPA1 antagonist HC-030031 [22,39,71-72]. TRPC4 and TRPC5 may be distinguished from other TRP channels by their potentiation by micromolar concentrations of La3+ [60,66]. Natural compounds, usually derived from plants, are ligands that are presumed to have evolved in the natural competition/symbiosis between plants and animals. Capsaicin is the well-known vanilloid in 'hot' peppers and seems to be relatively specific for TRPV1 [8,10].
PHYSIOLOGICAL FUNCTIONS
Mouse models and human genetics (see Channelopathies) have revealed diverse functions of TRP channels. Perhaps best characterized is the function of TRPV1 in primary afferent nociceptors. It is essential for the perception of noxious stimuli (heat, capsaicin) and contributes to the development of thermal hyperalgesia following inflammation [8,10]. Other examples for TRPs in somatosensation are TRPM8 that is activated by cooling or pharmacological agents evoking a 'cool' sensation and participates in the thermosensation of cold temperatures [5,13,38,51], or TRPA1 that is activated by pungent chemicals such as mustard oil (AITC), allicin, and cinnamaldehyde, and many reactive molecules. TRPA1 contributes to nociception [2-4,27].
TRPC2 is a pseudogene in humans, but in other mammals appears to be an ion channel localized to microvilli of the vomeronasal organ. It is required for normal sexual behavior in response to pheromones in mice [32,34,65].
Aside from their functions in sensory systems, TRP channels also participate in many more physiological processes for example within the renal, endocrine, cardiovascular, immune, or central nervous system. A comprehensive summary of physiological functions is provided for each channel in the corresponding tables.
TRP CHANNELOPATHIES
Mutations in TRP channel genes have been associated with inherited diseases in humans that affect organs and tissues in the renal, skeletal, skin epithelial, cardiovascular, or nervous system (reviewed by [42,74]). Molecular genetics identified various pathogenic mutations in TRP genes including nonsense mutations, splicing defects, frameshift deletions/insertions, missense or in-frame mutations. Both gain- and loss-of-function are linked to inherited diseases, but the pathophysiological mechanisms, i.e. how TRP channel dysfunction results in disease pathogenesis, are in most cases poorly understood. Since TRP channel signaling is generally tied to changes in intracellular Ca2+ levels, abnormal activity as a result of inherited mutations is likely to affect Ca2+-dependent cellular processes or cause toxic Ca2+ overload.
Among the TRP channels that are linked to hereditary channelopathies (see Table 1), TRPV4, TRPM6, and PKD2 (TRPP1) have the highest frequency of mutations [42,74]. More than 50 pathogenic mutations that cause different forms of skeletal dysplasia and peripheral neuropathies have been described for TRPV4 [45-46]. Recombinant expression studies suggest that TRPV4 mutations produce mostly gain-of-function phenotypes that are associated with increased basal Ca2+ levels [37,45]. How and why TRPV4 mutations cause different diseases is not well understood.
More than 35 mutations in TRPM6 are associated with autosomal recessive hypomagnesemia with secondary hypocalcemia 1 [30,42,62,74,77]. The disease is characterized by low serum Mg2+ and Ca2+ levels, presumably caused by dysregulated intestinal/renal Mg2+ reabsorption. How TRPM6 mutations result in defective Mg2+ reabsorption is unclear, but in recombinant expression systems mutant channels primarily exhibit loss-of-function [14,30,61,74].
PKD2 mutations are implicated in autosomal polycystic kidney disease (ADPKD, 15% of cases) [48]. A total of 278 germline mutations with varying clinical significance have been identified in PKD2 [53]. ADPKD is a late-onset disease that is characterized by progressive development of fluid-filled cysts in the kidney and other organs. Consistent with a PKD2 null mouse model [79], genetic defects in PKD2 are associated with loss-of-function phenotypes. The mechanisms linking dysfunctional PKD2 proteins to disease pathogenesis are not known.
Additional hereditary channelopathies have been described for TRPC6, TRPV3, TRPM1, TRPM4, TRPA1, and TRPML1 (see Table 1). The significance of mutations in TRPC3 and TRPM7 to disease pathogenesis in humans is as yet unclear. Detailed information is available through the OMIM database and also reviewed by [74].
Table 1. TRP channels and associated channelopathies. Based on OMIM, Online Mendelian Inheritance in Man database.
| TRP channel | Channelopathy | OMIM |
| TRPC6 | Glomerulosclerosis, focal segmental, 2 | 603965 |
| TRPV3 | Olmsted Syndrome | 614594 |
| TRPV4 | Brachyolmia type 3 | 113500 |
| Digital arthropathy-brachydactyly, familial | 606835 | |
| Hereditary motor and sensory neuropathy, type IIc | 606071 | |
| Metatropic dysplasia | 156530 | |
| Parastremmatic dwarfism | 168400 | |
| Scapuloperoneal spinal muscular atrophy | 181405 | |
| SED, Maroteaux type | 184095 | |
| Spinal muscular atrophy, distal, congenital nonprogressive | 600175 | |
| Spondylometaphyseal dysplasia, Kozlowski type | 184252 | |
| TRPM1 | Night blindness, congenital stationary (complete), 1C, autosomal recessive | 613216 |
| TRPM4 | Progressive familial heart block, type IB | 604559 |
| TRPM6 | Hypomagnesemia 1, intestinal | 602014 |
| TRPA1 | Episodic pain syndrome, familial | 615040 |
| TRPML1 | Mucolipidosis IV | 252650 |
| PKD2 (TRPP1) | Autosomal dominant polycystic kidney disease | 613095 |
References
1. Ambudkar IS, Ong HL. (2007) Organization and function of TRPC channelosomes. Pflugers Arch, 455 (2): 187-200. [PMID:17486362]
2. Bandell M, Story GM, Hwang SW, Viswanath V, Eid SR, Petrus MJ, Earley TJ, Patapoutian A. (2004) Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron, 41 (6): 849-57. [PMID:15046718]
3. Bautista DM, Jordt SE, Nikai T, Tsuruda PR, Read AJ, Poblete J, Yamoah EN, Basbaum AI, Julius D. (2006) TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell, 124 (6): 1269-82. [PMID:16564016]
4. Bautista DM, Movahed P, Hinman A, Axelsson HE, Sterner O, Högestätt ED, Julius D, Jordt SE, Zygmunt PM. (2005) Pungent products from garlic activate the sensory ion channel TRPA1. Proc Natl Acad Sci USA, 102 (34): 12248-52. [PMID:16103371]
5. Bautista DM, Siemens J, Glazer JM, Tsuruda PR, Basbaum AI, Stucky CL, Jordt SE, Julius D. (2007) The menthol receptor TRPM8 is the principal detector of environmental cold. Nature, 448 (7150): 204-8. [PMID:17538622]
6. Bork P. (1993) Hundreds of ankyrin-like repeats in functionally diverse proteins: mobile modules that cross phyla horizontally?. Proteins, 17 (4): 363-74. [PMID:8108379]
7. Cao E, Liao M, Cheng Y, Julius D. (2013) TRPV1 structures in distinct conformations reveal activation mechanisms. Nature, 504 (7478): 113-8. [PMID:24305161]
8. Caterina MJ, Leffler A, Malmberg AB, Martin WJ, Trafton J, Petersen-Zeitz KR, Koltzenburg M, Basbaum AI, Julius D. (2000) Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science, 288 (5464): 306-13. [PMID:10764638]
9. Caterina MJ, Rosen TA, Tominaga M, Brake AJ, Julius D. (1999) A capsaicin-receptor homologue with a high threshold for noxious heat. Nature, 398 (6726): 436-41. [PMID:10201375]
10. Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D. (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature, 389 (6653): 816-24. [PMID:9349813]
11. Catterall WA. (2010) Ion channel voltage sensors: structure, function, and pathophysiology. Neuron, 67 (6): 915-28. [PMID:20869590]
12. Cheng KT, Ong HL, Liu X, Ambudkar IS. (2011) Contribution of TRPC1 and Orai1 to Ca(2+) entry activated by store depletion. Adv Exp Med Biol, 704: 435-49. [PMID:21290310]
13. Chuang HH, Neuhausser WM, Julius D. (2004) The super-cooling agent icilin reveals a mechanism of coincidence detection by a temperature-sensitive TRP channel. Neuron, 43 (6): 859-69. [PMID:15363396]
14. Chubanov V, Waldegger S, Mederos y Schnitzler M, Vitzthum H, Sassen MC, Seyberth HW, Konrad M, Gudermann T. (2004) Disruption of TRPM6/TRPM7 complex formation by a mutation in the TRPM6 gene causes hypomagnesemia with secondary hypocalcemia. Proc Natl Acad Sci USA, 101 (9): 2894-9. [PMID:14976260]
15. Clapham DE. (2003) TRP channels as cellular sensors. Nature, 426 (6966): 517-24. [PMID:14654832]
16. Clapham DE. (2015) Structural biology: Pain-sensing TRPA1 channel resolved. Nature, 520 (7548): 439-41. [PMID:25855290]
17. Clapham DE, Garbers DL. (2005) International Union of Pharmacology. L. Nomenclature and structure-function relationships of CatSper and two-pore channels. Pharmacol Rev, 57 (4): 451-4. [PMID:16382101]
18. Clapham DE, Miller C. (2011) A thermodynamic framework for understanding temperature sensing by transient receptor potential (TRP) channels. Proc Natl Acad Sci U S A, 108 (49): 19492-7. [PMID:22109551]
19. Coste B, Mathur J, Schmidt M, Earley TJ, Ranade S, Petrus MJ, Dubin AE, Patapoutian A. (2010) Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science, 330 (6000): 55-60. [PMID:20813920]
20. Coste B, Xiao B, Santos JS, Syeda R, Grandl J, Spencer KS, Kim SE, Schmidt M, Mathur J, Dubin AE et al.. (2012) Piezo proteins are pore-forming subunits of mechanically activated channels. Nature, 483 (7388): 176-81. [PMID:22343900]
21. Erler I, Hirnet D, Wissenbach U, Flockerzi V, Niemeyer BA. (2004) Ca2+-selective transient receptor potential V channel architecture and function require a specific ankyrin repeat. J Biol Chem, 279 (33): 34456-63. [PMID:15192090]
22. Everaerts W, Zhen X, Ghosh D, Vriens J, Gevaert T, Gilbert JP, Hayward NJ, McNamara CR, Xue F, Moran MM et al.. (2010) Inhibition of the cation channel TRPV4 improves bladder function in mice and rats with cyclophosphamide-induced cystitis. Proc Natl Acad Sci USA, 107 (44): 19084-9. [PMID:20956320]
23. Feng Q. (2014) Temperature sensing by thermal TRP channels: thermodynamic basis and molecular insights. Curr Top Membr, 74: 19-50. [PMID:25366232]
24. Gees M, Owsianik G, Nilius B, Voets T. (2012) TRP channels. Compr Physiol, 2 (1): 563-608. [PMID:23728980]
25. Guler AD, Lee H, Iida T, Shimizu I, Tominaga M, Caterina M. (2002) Heat-evoked activation of the ion channel, TRPV4. J Neurosci, 22 (15): 6408-14. [PMID:12151520]
26. Huynh KW, Cohen MR, Jiang J, Samanta A, Lodowski DT, Zhou ZH, Moiseenkova-Bell VY. (2016) Structure of the full-length TRPV2 channel by cryo-EM. Nat Commun, 7: 11130. [PMID:27021073]
27. Jordt SE, Bautista DM, Chuang HH, McKemy DD, Zygmunt PM, Högestätt ED, Meng ID, Julius D. (2004) Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature, 427 (6971): 260-5. [PMID:14712238]
28. Karashima Y, Talavera K, Everaerts W, Janssens A, Kwan KY, Vennekens R, Nilius B, Voets T. (2009) TRPA1 acts as a cold sensor in vitro and in vivo. Proc Natl Acad Sci USA, 106 (4): 1273-8. [PMID:19144922]
29. Kawashima Y, Géléoc GS, Kurima K, Labay V, Lelli A, Asai Y, Makishima T, Wu DK, Della Santina CC, Holt JR et al.. (2011) Mechanotransduction in mouse inner ear hair cells requires transmembrane channel-like genes. J Clin Invest, 121 (12): 4796-809. [PMID:22105175]
30. Lainez S, Schlingmann KP, van der Wijst J, Dworniczak B, van Zeeland F, Konrad M, Bindels RJ, Hoenderop JG. (2014) New TRPM6 missense mutations linked to hypomagnesemia with secondary hypocalcemia. Eur J Hum Genet, 22 (4): 497-504. [PMID:23942199]
31. Levina N, Tötemeyer S, Stokes NR, Louis P, Jones MA, Booth IR. (1999) Protection of Escherichia coli cells against extreme turgor by activation of MscS and MscL mechanosensitive channels: identification of genes required for MscS activity. EMBO J, 18 (7): 1730-7. [PMID:10202137]
32. Leypold BG, Yu CR, Leinders-Zufall T, Kim MM, Zufall F, Axel R. (2002) Altered sexual and social behaviors in trp2 mutant mice. Proc Natl Acad Sci U S A, 99 (9): 6376-81. [PMID:11972034]
33. Liao M, Cao E, Julius D, Cheng Y. (2013) Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature, 504 (7478): 107-12. [PMID:24305160]
34. Liman ER, Corey DP, Dulac C. (1999) TRP2: a candidate transduction channel for mammalian pheromone sensory signaling. Proc Natl Acad Sci USA, 96 (10): 5791-6. [PMID:10318963]
35. Long SB, Campbell EB, Mackinnon R. (2005) Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science, 309 (5736): 897-903. [PMID:16002581]
36. Long SB, Tao X, Campbell EB, MacKinnon R. (2007) Atomic structure of a voltage-dependent K+ channel in a lipid membrane-like environment. Nature, 450 (7168): 376-82. [PMID:18004376]
37. Loukin S, Su Z, Kung C. (2011) Increased basal activity is a key determinant in the severity of human skeletal dysplasia caused by TRPV4 mutations. PLoS One, 6 (5): e19533. [PMID:21573172]
38. McKemy DD, Neuhausser WM, Julius D. (2002) Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature, 416 (6876): 52-8. [PMID:11882888]
39. McNamara CR, Mandel-Brehm J, Bautista DM, Siemens J, Deranian KL, Zhao M, Hayward NJ, Chong JA, Julius D, Moran MM et al.. (2007) TRPA1 mediates formalin-induced pain. Proc Natl Acad Sci USA, 104 (33): 13525-30. [PMID:17686976]
40. Montell C, Rubin GM. (1989) Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction. Neuron, 2 (4): 1313-23. [PMID:2516726]
41. Nadler MJ, Hermosura MC, Inabe K, Perraud AL, Zhu Q, Stokes AJ, Kurosaki T, Kinet JP, Penner R, Scharenberg AM et al.. (2001) LTRPC7 is a Mg.ATP-regulated divalent cation channel required for cell viability. Nature, 411 (6837): 590-5. [PMID:11385574]
42. Nilius B, Owsianik G. (2010) Transient receptor potential channelopathies. Pflugers Arch, 460 (2): 437-50. [PMID:20127491]
43. Nilius B, Owsianik G, Voets T. (2008) Transient receptor potential channels meet phosphoinositides. EMBO J, 27 (21): 2809-16. [PMID:18923420]
44. Nilius B, Talavera K, Owsianik G, Prenen J, Droogmans G, Voets T. (2005) Gating of TRP channels: a voltage connection?. J Physiol (Lond.), 567 (Pt 1): 35-44. [PMID:15878939]
45. Nilius B, Voets T. (2013) The puzzle of TRPV4 channelopathies. EMBO Rep, 14 (2): 152-63. [PMID:23306656]
46. Nishimura G, Lausch E, Savarirayan R, Shiba M, Spranger J, Zabel B, Ikegawa S, Superti-Furga A, Unger S. (2012) TRPV4-associated skeletal dysplasias. Am J Med Genet C Semin Med Genet, 160C (3): 190-204. [PMID:22791502]
47. Oancea E, Vriens J, Brauchi S, Jun J, Splawski I, Clapham DE. (2009) TRPM1 forms ion channels associated with melanin content in melanocytes. Sci Signal, 2 (70): ra21. [PMID:19436059]
48. Ong AC, Harris PC. (2015) A polycystin-centric view of cyst formation and disease: the polycystins revisited. Kidney Int, 88 (4): 699-710. [PMID:26200945]
49. Paulsen CE, Armache JP, Gao Y, Cheng Y, Julius D. (2015) Structure of the TRPA1 ion channel suggests regulatory mechanisms. Nature, 520 (7548): 511-7. [PMID:25855297]
50. Pedersen SF, Owsianik G, Nilius B. (2005) TRP channels: an overview. Cell Calcium, 38 (3-4): 233-52. [PMID:16098585]
51. Peier AM, Moqrich A, Hergarden AC, Reeve AJ, Andersson DA, Story GM, Earley TJ, Dragoni I, McIntyre P, Bevan S et al.. (2002) A TRP channel that senses cold stimuli and menthol. Cell, 108 (5): 705-15. [PMID:11893340]
52. Peier AM, Reeve AJ, Andersson DA, Moqrich A, Earley TJ, Hergarden AC, Story GM, Colley S, Hogenesch JB, McIntyre P et al.. (2002) A heat-sensitive TRP channel expressed in keratinocytes. Science, 296 (5575): 2046-9. [PMID:12016205]
53. PKD Foundation. Autosomal Dominant Polycystic Kidney Disease Mutation Database: PKDB. Accessed on 24/08/2021. Modified on 24/08/2021. pkdb.mayo.edu, http://pkdb.mayo.edu
54. Plant TD. (2014) TRPs in mechanosensing and volume regulation. Handb Exp Pharmacol, 223: 743-66. [PMID:24961968]
55. Potier M, Trebak M. (2008) New developments in the signaling mechanisms of the store-operated calcium entry pathway. Pflugers Arch, 457 (2): 405-15. [PMID:18536932]
56. Ramsey IS, Delling M, Clapham DE. (2006) An introduction to TRP channels. Annu Rev Physiol, 68: 619-47. [PMID:16460286]
57. Rohacs T, Nilius B. (2007) Regulation of transient receptor potential (TRP) channels by phosphoinositides. Pflugers Arch, 455 (1): 157-68. [PMID:17479281]
58. Runnels LW, Yue L, Clapham DE. (2001) TRP-PLIK, a bifunctional protein with kinase and ion channel activities. Science, 291 (5506): 1043-7. [PMID:11161216]
59. Salido GM, Sage SO, Rosado JA. (2009) TRPC channels and store-operated Ca(2+) entry. Biochim Biophys Acta, 1793 (2): 223-30. [PMID:19061922]
60. Schaefer M, Plant TD, Obukhov AG, Hofmann T, Gudermann T, Schultz G. (2000) Receptor-mediated regulation of the nonselective cation channels TRPC4 and TRPC5. J Biol Chem, 275 (23): 17517-26. [PMID:10837492]
61. Schlingmann KP, Sassen MC, Weber S, Pechmann U, Kusch K, Pelken L, Lotan D, Syrrou M, Prebble JJ, Cole DE et al.. (2005) Novel TRPM6 mutations in 21 families with primary hypomagnesemia and secondary hypocalcemia. J Am Soc Nephrol, 16 (10): 3061-9. [PMID:16107578]
62. Schlingmann KP, Weber S, Peters M, Niemann Nejsum L, Vitzthum H, Klingel K, Kratz M, Haddad E, Ristoff E, Dinour D et al.. (2002) Hypomagnesemia with secondary hypocalcemia is caused by mutations in TRPM6, a new member of the TRPM gene family. Nat Genet, 31 (2): 166-70. [PMID:12032568]
63. Smith GD, Gunthorpe MJ, Kelsell RE, Hayes PD, Reilly P, Facer P, Wright JE, Jerman JC, Walhin JP, Ooi L et al.. (2002) TRPV3 is a temperature-sensitive vanilloid receptor-like protein. Nature, 418 (6894): 186-90. [PMID:12077606]
64. Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, Earley TJ, Hergarden AC, Andersson DA, Hwang SW et al.. (2003) ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell, 112 (6): 819-29. [PMID:12654248]
65. Stowers L, Holy TE, Meister M, Dulac C, Koentges G. (2002) Loss of sex discrimination and male-male aggression in mice deficient for TRP2. Science, 295 (5559): 1493-500. [PMID:11823606]
66. Strübing C, Krapivinsky G, Krapivinsky L, Clapham DE. (2001) TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron, 29 (3): 645-55. [PMID:11301024]
67. Strübing C, Krapivinsky G, Krapivinsky L, Clapham DE. (2003) Formation of novel TRPC channels by complex subunit interactions in embryonic brain. J Biol Chem, 278 (40): 39014-9. [PMID:12857742]
68. Sukharev S. (2002) Purification of the small mechanosensitive channel of Escherichia coli (MscS): the subunit structure, conduction, and gating characteristics in liposomes. Biophys J, 83 (1): 290-8. [PMID:12080120]
69. Sukharev SI, Blount P, Martinac B, Blattner FR, Kung C. (1994) A large-conductance mechanosensitive channel in E. coli encoded by mscL alone. Nature, 368 (6468): 265-8. [PMID:7511799]
70. Talavera K, Yasumatsu K, Voets T, Droogmans G, Shigemura N, Ninomiya Y, Margolskee RF, Nilius B. (2005) Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature, 438 (7070): 1022-5. [PMID:16355226]
71. Thorneloe KS, Cheung M, Bao W, Alsaid H, Lenhard S, Jian MY, Costell M, Maniscalco-Hauk K, Krawiec JA, Olzinski A et al.. (2012) An orally active TRPV4 channel blocker prevents and resolves pulmonary edema induced by heart failure. Sci Transl Med, 4 (159): 159ra148. [PMID:23136043]
72. Thorneloe KS, Sulpizio AC, Lin Z, Figueroa DJ, Clouse AK, McCafferty GP, Chendrimada TP, Lashinger ES, Gordon E, Evans L et al.. (2008) N-((1S)-1-{[4-((2S)-2-{[(2,4-dichlorophenyl)sulfonyl]amino}-3-hydroxypropanoyl)-1-piperazinyl]carbonyl}-3-methylbutyl)-1-benzothiophene-2-carboxamide (GSK1016790A), a novel and potent transient receptor potential vanilloid 4 channel agonist induces urinary bladder contraction and hyperactivity: Part I. J Pharmacol Exp Ther, 326 (2): 432-42. [PMID:18499743]
73. Tóth B, Iordanov I, Csanády L. (2015) Ruling out pyridine dinucleotides as true TRPM2 channel activators reveals novel direct agonist ADP-ribose-2'-phosphate. J Gen Physiol, 145 (5): 419-30. [PMID:25918360]
74. Tóth BI, Nilius B. (2015) Transient Receptor Potential Dysfunctions in Hereditary Diseases: TRP Channelopathies and Beyond. In TRP Channels as Therapeutic Target Edited by Szallasi A (Elsevier Inc) 13-33. DOI: 10.1016/C2013-0-09923-9 [ISBN:9780124200241]
75. Venkatachalam K, Montell C. (2007) TRP channels. Annu Rev Biochem, 76: 387-417. [PMID:17579562]
76. Vriens J, Owsianik G, Hofmann T, Philipp SE, Stab J, Chen X, Benoit M, Xue F, Janssens A, Kerselaers S et al.. (2011) TRPM3 is a nociceptor channel involved in the detection of noxious heat. Neuron, 70 (3): 482-94. [PMID:21555074]
77. Walder RY, Landau D, Meyer P, Shalev H, Tsolia M, Borochowitz Z, Boettger MB, Beck GE, Englehardt RK, Carmi R et al.. (2002) Mutation of TRPM6 causes familial hypomagnesemia with secondary hypocalcemia. Nat Genet, 31 (2): 171-4. [PMID:12032570]
78. Watanabe H, Vriens J, Suh SH, Benham CD, Droogmans G, Nilius B. (2002) Heat-evoked activation of TRPV4 channels in a HEK293 cell expression system and in native mouse aorta endothelial cells. J Biol Chem, 277 (49): 47044-51. [PMID:12354759]
79. Wu G, Markowitz GS, Li L, D'Agati VD, Factor SM, Geng L, Tibara S, Tuchman J, Cai Y, Park JH et al.. (2000) Cardiac defects and renal failure in mice with targeted mutations in Pkd2. Nat Genet, 24 (1): 75-8. [PMID:10615132]
80. Wu LJ, Sweet TB, Clapham DE. (2010) International Union of Basic and Clinical Pharmacology. LXXVI. Current progress in the mammalian TRP ion channel family. Pharmacol Rev, 62 (3): 381-404. [PMID:20716668]
81. Xu H, Ramsey IS, Kotecha SA, Moran MM, Chong JA, Lawson D, Ge P, Lilly J, Silos-Santiago I, Xie Y et al.. (2002) TRPV3 is a calcium-permeable temperature-sensitive cation channel. Nature, 418 (6894): 181-6. [PMID:12077604]
82. Xu H, Ren D. (2015) Lysosomal physiology. Annu Rev Physiol, 77: 57-80. [PMID:25668017]
83. Yuan JP, Kim MS, Zeng W, Shin DM, Huang G, Worley PF, Muallem S. (2009) TRPC channels as STIM1-regulated SOCs. Channels (Austin), 3 (4): 221-5. [PMID:19574740]
84. Zimmermann K, Lennerz JK, Hein A, Link AS, Kaczmarek JS, Delling M, Uysal S, Pfeifer JD, Riccio A, Clapham DE. (2011) Transient receptor potential cation channel, subfamily C, member 5 (TRPC5) is a cold-transducer in the peripheral nervous system. Proc Natl Acad Sci USA, 108 (44): 18114-9. [PMID:22025699]
85. Zubcevic L, Herzik Jr MA, Chung BC, Liu Z, Lander GC, Lee SY. (2016) Cryo-electron microscopy structure of the TRPV2 ion channel. Nat Struct Mol Biol, 23 (2): 180-186. [PMID:26779611]