|
Synonyms: J3.333.591A | LFF 571 | LFF-571
Comment: LFF571 is a macrocyclic thiopetide with activity against Gram-positive bacteria and derived from the naturally occurring antibacterial GE2270 A [ 2]. Functionally, it inhibits bacterial protein synthesis by targeting elongation factor Tu (EF-Tu) [ 3]. Note that the structure shown here matches the CAS-assigned structure. The PubChem link, provided in the table below, is to their standardized chemical structure (CID 42638236), although this is not an exact structural match.
|
|
2D Structure
|
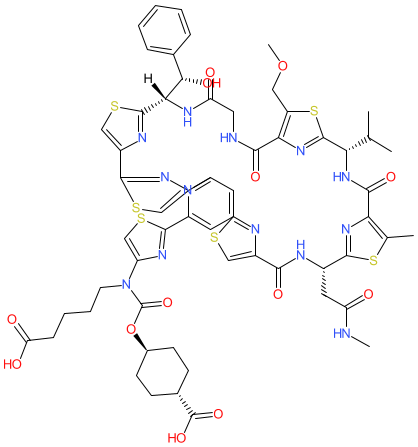
|
SMILES / InChI / InChIKey
|
|
|
Canonical SMILES
|
CC(C)[C@H]1C2=NC(=C(COC)S2)C(=O)NCC(=O)N[C@@]([H])([C@H](C3=CC=CC=C3)O)C4=NC(=CS4)C5=NC(=CS5)C6=NC(=CC=C6C7=NC(=CS7)C(=O)N[C@@H](CC(=O)NC)C8=NC(=C(C)S8)C(=O)N1)C9=NC(=CS9)N(CCCCC(=O)O)C(=O)O[C@H]%10CC[C@@H](CC%10)C(=O)O
|
|
Isomeric SMILES
|
O=C1C=2N=C(C=3C(C=4N=C(SC4)C=5N=C(SC5)[C@]([C@@H](O)C6=CC=CC=C6)(NC(=O)CNC(=O)C7=C(COC)SC([C@H](C(C)C)NC(=O)C=8N=C([C@H](CC(NC)=O)N1)SC8C)=N7)[H])=NC(=CC3)C9=NC(N(C(O[C@@H]%10CC[C@@H](C(O)=O)CC%10)=O)CCCCC(O)=O)=CS9)SC2
|
|
InChI
|
InChI=1S/C60H63N13O13S6/c1-28(2)44-58-72-47(39(92-58)23-85-5)51(80)62-22-42(75)69-48(49(78)30-11-7-6-8-12-30)57-67-38(26-89-57)55-65-36(24-88-55)46-33(53-66-37(25-87-53)50(79)64-35(21-41(74)61-4)56-71-45(29(3)91-56)52(81)70-44)18-19-34(63-46)54-68-40(27-90-54)73(20-10-9-13-43(76)77)60(84)86-32-16-14-31(15-17-32)59(82)83/h6-8,11-12,18-19,24-28,31-32,35,44,48-49,78H,9-10,13-17,20-23H2,1-5H3,(H,61,74)(H,62,80)(H,64,79)(H,69,75)(H,70,81)(H,76,77)(H,82,83)/t31-,32-,35-,44-,48-,49-/m0/s1
|
|
InChI Key
|
GNLYKLDXQZHYTR-LMOGNUDZSA-N
|
Generated using the Chemistry Development Kit (CDK) (Willighagen EL et al. Journal of Cheminformatics vol. 9:33. 2017, doi:10.1186/s13321-017-0220-4; https://cdk.github.io/)
|
|


