|
Synonyms: A-16686A2 | MDL 62,198
Comment: Ramoplanin is a glycolipodepsipeptide antibacterial with Gram-positive activity, originally isolated from the culture broth of Actinoplanes strain ATCC 33076 as a mixture of three components (ramoplanin A1, A2, and A3) [ 1, 3]. We show the structure for ramoplanin A2 which is the major component. Note that the INN describes the mixture. As with other natural products, there is ambiguity in the precise stereochemisrty presented across web resources e.g., CHEMBL3989605 shows an alternate structure.
|
|
2D Structure
|
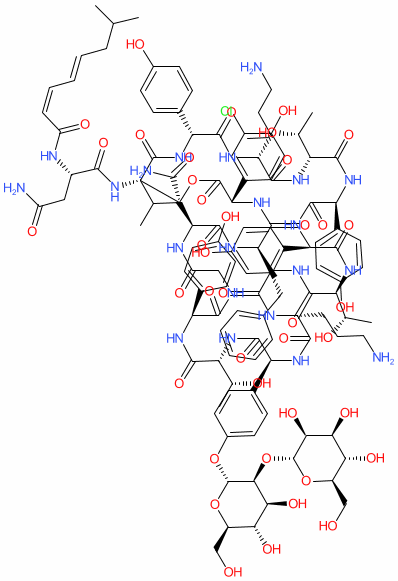
|
SMILES / InChI / InChIKey
|
|
|
Canonical SMILES
|
CC(C)C/C=C/C=C\C(=O)N[C@@H](CC(=O)N)C(=O)N[C@H]1[C@@H](C(=O)N)OC(=O)[C@H](C2=CC=C(C(=C2)Cl)O)NC(=O)[C@@H](C)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C3=CC=C(C=C3)O)NC(=O)[C@@H]([C@@H](C)O)NC(=O)[C@H](C4=CC=C(C=C4)O[C@@H]5[C@H]([C@H]([C@@H]([C@@H](CO)O5)O)O)O[C@@H]6[C@H]([C@H]([C@@H]([C@@H](CO)O6)O)O)O)NC(=O)[C@@H](CCCN)NC(=O)[C@H](CC7=CC=CC=C7)NC(=O)[C@H]([C@H](C)O)NC(=O)[C@@H](C8=CC=C(C=C8)O)NC(=O)[C@H](C9=CC=C(C=C9)O)NC(=O)[C@@H]([C@@H](C)O)NC(=O)[C@@H](CCCN)NC(=O)[C@@H](C%10=CC=C(C=C%10)O)NC1=O
|
|
Isomeric SMILES
|
C[C@@H]1C(=O)N[C@H](C(=O)O[C@@H]([C@@H](C(=O)N[C@@H](C(=O)N[C@@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)NCC(=O)N[C@H](C(=O)N1)CC(C)C)C2=CC=C(C=C2)O)[C@@H](C)O)C3=CC=C(C=C3)O[C@@H]4[C@H]([C@H]([C@@H]([C@H](O4)CO)O)O)O[C@@H]5[C@H]([C@H]([C@@H]([C@H](O5)CO)O)O)O)CCCN)CC6=CC=CC=C6)[C@H](C)O)C7=CC=C(C=C7)O)C8=CC=C(C=C8)O)[C@@H](C)O)CCCN)C9=CC=C(C=C9)O)NC(=O)[C@H](CC(=O)N)NC(=O)/C=C\C=C\CC(C)C)C(=O)N)C%10=CC(=C(C=C%10)O)Cl
|
|
InChI
|
InChI=1S/C119H154ClN21O40/c1-54(2)17-11-9-14-22-82(153)127-77(50-81(123)152)107(166)141-93-99(101(124)160)180-117(176)92(66-33-44-78(151)72(120)49-66)140-102(161)56(5)126-105(164)75(47-55(3)4)128-83(154)51-125-108(167)87(61-23-34-67(147)35-24-61)136-111(170)86(59(8)146)134-113(172)89(65-31-42-71(43-32-65)177-119-100(97(158)95(156)80(53-143)179-119)181-118-98(159)96(157)94(155)79(52-142)178-118)135-104(163)73(20-15-45-121)129-106(165)76(48-60-18-12-10-13-19-60)131-109(168)84(57(6)144)133-114(173)90(63-27-38-69(149)39-28-63)138-115(174)91(64-29-40-70(150)41-30-64)137-110(169)85(58(7)145)132-103(162)74(21-16-46-122)130-112(171)88(139-116(93)175)62-25-36-68(148)37-26-62/h9-14,18-19,22-44,49,54-59,73-77,79-80,84-100,118-119,142-151,155-159H,15-17,20-21,45-48,50-53,121-122H2,1-8H3,(H2,123,152)(H2,124,160)(H,125,167)(H,126,164)(H,127,153)(H,128,154)(H,129,165)(H,130,171)(H,131,168)(H,132,162)(H,133,173)(H,134,172)(H,135,163)(H,136,170)(H,137,169)(H,138,174)(H,139,175)(H,140,161)(H,141,166)/b11-9+,22-14-/t56-,57+,58-,59-,73-,74-,75+,76+,77+,79-,80-,84+,85-,86-,87+,88-,89+,90-,91+,92+,93+,94-,95-,96+,97+,98+,99+,100+,118-,119+/m1/s1
|
|
InChI Key
|
KGZHFKDNSAEOJX-WIFQYKSHSA-N
|
Generated using the Chemistry Development Kit (CDK) (Willighagen EL et al. Journal of Cheminformatics vol. 9:33. 2017, doi:10.1186/s13321-017-0220-4; https://cdk.github.io/)
|
|






