
Top ▲

GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Gene and Protein Information  |
||||||
| class A G protein-coupled receptor | ||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | 7 | 425 | 5q13.3 | F2R | coagulation factor II thrombin receptor | |
| Mouse | 7 | 430 | 13 50.21 cM | F2r | coagulation factor II thrombin receptor | |
| Rat | 7 | 432 | 2q12 | F2r | coagulation factor II (thrombin) receptor | |
Previous and Unofficial Names  |
| protease-activated receptor 1 | coagulation factor II (thrombin) receptor |
Database Links  |
|
| Specialist databases | |
| GPCRdb | par1_human (Hs), par1_mouse (Mm), par1_rat (Rn) |
| Other databases | |
| Alphafold | P25116 (Hs), P30558 (Mm), P26824 (Rn) |
| ChEMBL Target | CHEMBL3974 (Hs), CHEMBL4523212 (Mm), CHEMBL6041 (Rn) |
| Ensembl Gene | ENSG00000181104 (Hs), ENSMUSG00000048376 (Mm), ENSRNOG00000048043 (Rn) |
| Entrez Gene | 2149 (Hs), 14062 (Mm), 25439 (Rn) |
| Human Protein Atlas | ENSG00000181104 (Hs) |
| KEGG Gene | hsa:2149 (Hs), mmu:14062 (Mm), rno:25439 (Rn) |
| OMIM | 187930 (Hs) |
| Pharos | P25116 (Hs) |
| RefSeq Nucleotide | NM_001992 (Hs), NM_010169 (Mm), NM_012950 (Rn) |
| RefSeq Protein | NP_001983 (Hs), NP_034299 (Mm), NP_037082 (Rn) |
| SynPHARM | 9403 (in complex with vorapaxar) |
| UniProtKB | P25116 (Hs), P30558 (Mm), P26824 (Rn) |
| Wikipedia | F2R (Hs) |
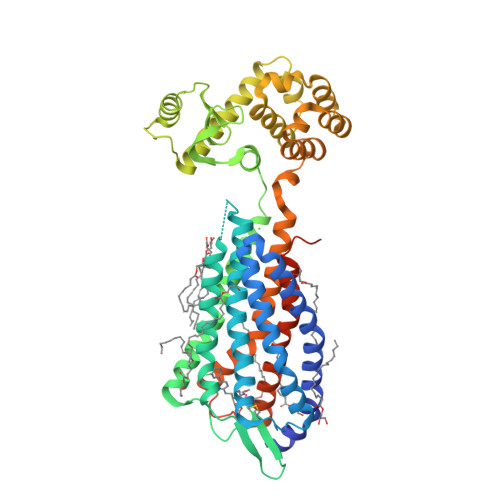
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||
Natural/Endogenous Ligands  |
| thrombin {Sp: Human} , thrombin {Sp: Mouse} , thrombin {Sp: Rat} |
| Agonist proteases (Human) |
| thrombin (F2, P00734), activated protein C (PROC, P04070), matrix metalloproteinase 1 (MMP1, P45452), matrix metalloproteinase 13 (MMP13, P45452) [9] |
Download all structure-activity data for this target as a CSV file 
| Agonists | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonists | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| PAR1 has been found to mediate the effects of neutrophil elastase (NE) on γδ T cell activation, as the PAR1 inhibitor RWJ-56110 abrogates NE (from neutrophils)-induced activation of these T cells [29]. We have therefore included PAR1 in the GtoImmuPdb. |
Primary Transduction Mechanisms 
|
|
| Transducer | Effector/Response |
|
Gi/Go family Gq/G11 family G12/G13 family |
|
| References: | |
Expression Datasets  |
|
|
Physiological Consequences of Altering Gene Expression 
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
||||||||||
|
Xenobiotics Influencing Gene Expression 
|
||||||||||
|
Phenotypes, Alleles and Disease Models 
|
Mouse data from MGI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Biologically Significant Variants 
|
||||||||
|
1. Ahn HS, Foster C, Boykow G, Arik L, Smith-Torhan A, Hesk D, Chatterjee M. (1997) Binding of a thrombin receptor tethered ligand analogue to human platelet thrombin receptor. Mol Pharmacol, 51 (2): 350-6. [PMID:9203642]
2. Ahn HS, Foster C, Boykow G, Stamford A, Manna M, Graziano M. (2000) Inhibition of cellular action of thrombin by N3-cyclopropyl-7-[[4-(1-methylethyl)phenyl]methyl]-7H-pyrrolo[3, 2-f]quinazoline-1,3-diamine (SCH 79797), a nonpeptide thrombin receptor antagonist. Biochem Pharmacol, 60 (10): 1425-34. [PMID:11020444]
3. Almonte AG, Hamill CE, Chhatwal JP, Wingo TS, Barber JA, Lyuboslavsky PN, David Sweatt J, Ressler KJ, White DA, Traynelis SF. (2007) Learning and memory deficits in mice lacking protease activated receptor-1. Neurobiol Learn Mem, 88 (3): 295-304. [PMID:17544303]
4. Almonte AG, Qadri LH, Sultan FA, Watson JA, Mount DJ, Rumbaugh G, Sweatt JD. (2013) Protease-activated receptor-1 modulates hippocampal memory formation and synaptic plasticity. J Neurochem, 124 (1): 109-22. [PMID:23113835]
5. Andrade-Gordon P, Maryanoff BE, Derian CK, Zhang HC, Addo MF, Darrow AL, Eckardt AJ, Hoekstra WJ, McComsey DF, Oksenberg D et al.. (1999) Design, synthesis, and biological characterization of a peptide-mimetic antagonist for a tethered-ligand receptor. Proc Natl Acad Sci USA, 96 (22): 12257-62. [PMID:10535908]
6. Antoniak S, Owens 3rd AP, Baunacke M, Williams JC, Lee RD, Weithäuser A, Sheridan PA, Malz R, Luyendyk JP, Esserman DA et al.. (2013) PAR-1 contributes to the innate immune response during viral infection. J Clin Invest, 123 (3): 1310-22. [PMID:23391721]
7. Aronovich A, Nur Y, Shezen E, Rosen C, Zlotnikov Klionsky Y, Milman I, Yarimi L, Hagin D, Rechavi G, Martinowitz U et al.. (2013) A novel role for factor VIII and thrombin/PAR1 in regulating hematopoiesis and its interplay with the bone structure. Blood, 122 (15): 2562-71. [PMID:23982175]
8. Atzori L, Lucattelli M, Scotton CJ, Laurent GJ, Bartalesi B, De Cunto G, Lunghi B, Chambers RC, Lungarella G. (2009) Absence of proteinase-activated receptor-1 signaling in mice confers protection from fMLP-induced goblet cell metaplasia. Am J Respir Cell Mol Biol, 41 (6): 680-7. [PMID:19307611]
9. Austin KM, Covic L, Kuliopulos A. (2013) Matrix metalloproteases and PAR1 activation. Blood, 121 (3): 431-9. [PMID:23086754]
10. Bourgognon JM, Schiavon E, Salah-Uddin H, Skrzypiec AE, Attwood BK, Shah RS, Patel SG, Mucha M, John Challiss RA, Forsythe ID et al.. (2013) Regulation of neuronal plasticity and fear by a dynamic change in PAR1-G protein coupling in the amygdala. Mol Psychiatry, 18 (10): 1136-45. [PMID:23032873]
11. Burda JE, Radulovic M, Yoon H, Scarisbrick IA. (2013) Critical role for PAR1 in kallikrein 6-mediated oligodendrogliopathy. Glia, 61 (9): 1456-70. [PMID:23832758]
12. Chackalamannil S, Wang Y, Greenlee WJ, Hu Z, Xia Y, Ahn HS, Boykow G, Hsieh Y, Palamanda J, Agans-Fantuzzi J et al.. (2008) Discovery of a novel, orally active himbacine-based thrombin receptor antagonist (SCH 530348) with potent antiplatelet activity. J Med Chem, 51 (11): 3061-4. [PMID:18447380]
13. Cheng Y, Xi G, Jin H, Keep RF, Feng J, Hua Y. (2014) Thrombin-induced cerebral hemorrhage: role of protease-activated receptor-1. Transl Stroke Res, 5 (4): 472-5. [PMID:24323711]
14. Guo H, Liu D, Gelbard H, Cheng T, Insalaco R, Fernández JA, Griffin JH, Zlokovic BV. (2004) Activated protein C prevents neuronal apoptosis via protease activated receptors 1 and 3. Neuron, 41 (4): 563-72. [PMID:14980205]
15. Hamill CE, Mannaioni G, Lyuboslavsky P, Sastre AA, Traynelis SF. (2009) Protease-activated receptor 1-dependent neuronal damage involves NMDA receptor function. Exp Neurol, 217 (1): 136-46. [PMID:19416668]
16. Hollenberg MD, Saifeddine M, al-Ani B, Kawabata A. (1997) Proteinase-activated receptors: structural requirements for activity, receptor cross-reactivity, and receptor selectivity of receptor-activating peptides. Can J Physiol Pharmacol, 75 (7): 832-41. [PMID:9315351]
17. Howell DC, Johns RH, Lasky JA, Shan B, Scotton CJ, Laurent GJ, Chambers RC. (2005) Absence of proteinase-activated receptor-1 signaling affords protection from bleomycin-induced lung inflammation and fibrosis. Am J Pathol, 166 (5): 1353-65. [PMID:15855637]
18. Jenkins RG, Su X, Su G, Scotton CJ, Camerer E, Laurent GJ, Davis GE, Chambers RC, Matthay MA, Sheppard D. (2006) Ligation of protease-activated receptor 1 enhances alpha(v)beta6 integrin-dependent TGF-beta activation and promotes acute lung injury. J Clin Invest, 116 (6): 1606-14. [PMID:16710477]
19. Junge CE, Sugawara T, Mannaioni G, Alagarsamy S, Conn PJ, Brat DJ, Chan PH, Traynelis SF. (2003) The contribution of protease-activated receptor 1 to neuronal damage caused by transient focal cerebral ischemia. Proc Natl Acad Sci USA, 100 (22): 13019-24. [PMID:14559973]
20. Nagai T, Ito M, Nakamichi N, Mizoguchi H, Kamei H, Fukakusa A, Nabeshima T, Takuma K, Yamada K. (2006) The rewards of nicotine: regulation by tissue plasminogen activator-plasmin system through protease activated receptor-1. J Neurosci, 26 (47): 12374-83. [PMID:17122062]
21. Nagai T, Nabeshima T, Yamada K. (2008) Basic and translational research on proteinase-activated receptors: regulation of nicotine reward by the tissue plasminogen activator (tPA) - plasmin system via proteinase-activated receptor 1. J Pharmacol Sci, 108 (4): 408-14. [PMID:19098386]
22. Patel YM, Lordkipanidzé M, Lowe GC, Nisar SP, Garner K, Stockley J, Daly ME, Mitchell M, Watson SP, Austin SK et al.. (2014) A novel mutation in the P2Y12 receptor and a function-reducing polymorphism in protease-activated receptor 1 in a patient with chronic bleeding. J Thromb Haemost, 12 (5): 716-25. [PMID:24612435]
23. Rajput PS, Lyden PD, Chen B, Lamb JA, Pereira B, Lamb A, Zhao L, Lei IF, Bai J. (2014) Protease activated receptor-1 mediates cytotoxicity during ischemia using in vivo and in vitro models. Neuroscience, 281: 229-40. [PMID:25261684]
24. Rullier A, Gillibert-Duplantier J, Costet P, Cubel G, Haurie V, Petibois C, Taras D, Dugot-Senant N, Deleris G, Bioulac-Sage P et al.. (2008) Protease-activated receptor 1 knockout reduces experimentally induced liver fibrosis. Am J Physiol Gastrointest Liver Physiol, 294 (1): G226-35. [PMID:17962354]
25. Schouten M, van't Veer C, Roelofs JJ, Levi M, van der Poll T. (2012) Protease-activated receptor-1 impairs host defense in murine pneumococcal pneumonia: a controlled laboratory study. Crit Care, 16 (6): R238. [PMID:23270594]
26. Sevastos J, Kennedy SE, Davis DR, Sam M, Peake PW, Charlesworth JA, Mackman N, Erlich JH. (2007) Tissue factor deficiency and PAR-1 deficiency are protective against renal ischemia reperfusion injury. Blood, 109 (2): 577-83. [PMID:16990608]
27. Smith SM, Judge HM, Peters G, Armstrong M, Dupont A, Gaussem P, Storey RF. (2005) PAR-1 genotype influences platelet aggregation and procoagulant responses in patients with coronary artery disease prior to and during clopidogrel therapy. Platelets, 16 (6): 340-5. [PMID:16194864]
28. Strompfová V, Lauková A, Cilik D. (2013) Synbiotic administration of canine-derived strain Lactobacillus fermentum CCM 7421 and inulin to healthy dogs. Can J Microbiol, 59 (5): 347-52. [PMID:23647348]
29. Towstyka NY, Shiromizu CM, Keitelman I, Sabbione F, Salamone GV, Geffner JR, Trevani AS, Jancic CC. (2018) Modulation of γδ T-cell activation by neutrophil elastase. Immunology, 153 (2): 225-237. [PMID:28888033]
30. Wang J, Jin H, Hua Y, Keep RF, Xi G. (2012) Role of protease-activated receptor-1 in brain injury after experimental global cerebral ischemia. Stroke, 43 (9): 2476-82. [PMID:22811450]
31. Wang Y, Zhao Z, Chow N, Ali T, Griffin JH, Zlokovic BV. (2013) Activated protein C analog promotes neurogenesis and improves neurological outcome after focal ischemic stroke in mice via protease activated receptor 1. Brain Res, 1507: 97-104. [PMID:23438513]
32. Wee JL, Chionh YT, Ng GZ, Harbour SN, Allison C, Pagel CN, Mackie EJ, Mitchell HM, Ferrero RL, Sutton P. (2010) Protease-activated receptor-1 down-regulates the murine inflammatory and humoral response to Helicobacter pylori. Gastroenterology, 138 (2): 573-82. [PMID:19706295]
33. Xie Q, Xi G, Gong Y, Keep R, Muraszko K, Hua Y. (2013) Protease activated receptor-1 and brain edema formation in glioma models. Acta Neurochir Suppl, 118: 191-4. [PMID:23564130]
34. Zhang C, Srinivasan Y, Arlow DH, Fung JJ, Palmer D, Zheng Y, Green HF, Pandey A, Dror RO, Shaw DE et al.. (2012) High-resolution crystal structure of human protease-activated receptor 1. Nature, 492 (7429): 387-92. [PMID:23222541]
35. Zhou QB, Duan CZ, Jia Q, Liu P, Li LY. (2014) Baicalin attenuates focal cerebral ischemic reperfusion injury by inhibition of protease-activated receptor-1 and apoptosis. Chin J Integr Med, 20 (2): 116-22. [PMID:23504578]