|
|
|
2D Structure
|
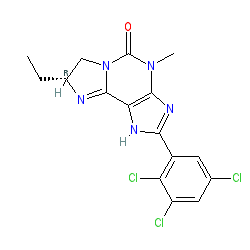
|
Physico-chemical Properties
|
|
|
Hydrogen bond acceptors
|
3
|
|
Hydrogen bond donors
|
1
|
|
Rotatable bonds
|
2
|
|
Topological polar surface area
|
67.97
|
|
Molecular weight
|
397.03
|
|
XLogP
|
5.88
|
|
No. Lipinski's rules broken
|
1
|
Generated using the Chemistry Development Kit (CDK) (Willighagen EL et al. Journal of Cheminformatics vol. 9:33. 2017, doi:10.1186/s13321-017-0220-4; https://cdk.github.io/)
|
SMILES / InChI / InChIKey
|
|
|
Canonical SMILES
|
CCC1N=c2n(C1)c(=O)n(c1c2[nH]c(n1)c1cc(Cl)cc(c1Cl)Cl)C
|
|
Isomeric SMILES
|
CC[C@H]1N=c2n(C1)c(=O)n(c1c2[nH]c(n1)c1cc(Cl)cc(c1Cl)Cl)C
|
|
InChI
|
InChI=1S/C16H14Cl3N5O/c1-3-8-6-24-15(20-8)12-14(23(2)16(24)25)22-13(21-12)9-4-7(17)5-10(18)11(9)19/h4-5,8H,3,6H2,1-2H3,(H,21,22)/t8-/m1/s1
|
|
InChI Key
|
YYDHUJWLNPIBDS-MRVPVSSYSA-N
|
Generated using the Chemistry Development Kit (CDK) (Willighagen EL et al. Journal of Cheminformatics vol. 9:33. 2017, doi:10.1186/s13321-017-0220-4; https://cdk.github.io/)
|
|

