
Top ▲

GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Target not currently curated in GtoImmuPdb
Target id: 1944
Nomenclature: bromodomain containing 2
Abbreviated Name: BRD2
Family: Bromodomain kinase (BRDK) family
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 801 | 6p21.32 | BRD2 | bromodomain containing 2 | |
| Mouse | - | 798 | 17 17.98 cM | Brd2 | bromodomain containing 2 | |
| Rat | - | 798 | 20p12 | Brd2 | bromodomain containing 2 | |
Previous and Unofficial Names  |
| FSRG1 | NAT | Protein RING3 |
Database Links  |
|
| Alphafold | P25440 (Hs), Q7JJ13 (Mm), Q6MGA9 (Rn) |
| BRENDA | 2.7.11.1 |
| CATH/Gene3D | 1.20.920.10 |
| ChEMBL Target | CHEMBL1293289 (Hs) |
| Ensembl Gene | ENSG00000204256 (Hs), ENSMUSG00000024335 (Mm), ENSRNOG00000000461 (Rn) |
| Entrez Gene | 6046 (Hs), 14312 (Mm), 294276 (Rn) |
| Human Protein Atlas | ENSG00000204256 (Hs) |
| KEGG Enzyme | 2.7.11.1 |
| KEGG Gene | hsa:6046 (Hs), mmu:14312 (Mm), rno:294276 (Rn) |
| OMIM | 601540 (Hs) |
| Pharos | P25440 (Hs) |
| RefSeq Nucleotide | NM_001113182 (Hs), NM_001025387 (Mm), NM_212495 (Rn) |
| RefSeq Protein | NP_001106653 (Hs), NP_001191902 (Mm), NP_997660 (Rn) |
| SynPHARM |
80624 (in complex with ET bromodomain inhibitor) 79907 (in complex with GW841819X) 79914 (in complex with I-BET151) 79905 (in complex with I-BET-762) 80623 (in complex with ME bromodomain inhibitor) 82687 (in complex with RVX 208) |
| UniProtKB | P25440 (Hs), Q7JJ13 (Mm), Q6MGA9 (Rn) |
| Wikipedia | BRD2 (Hs) |
Selected 3D Structures  |
|||||||||||||
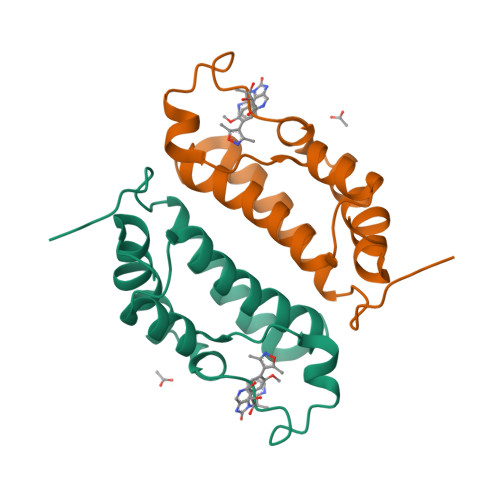
|
|
||||||||||||
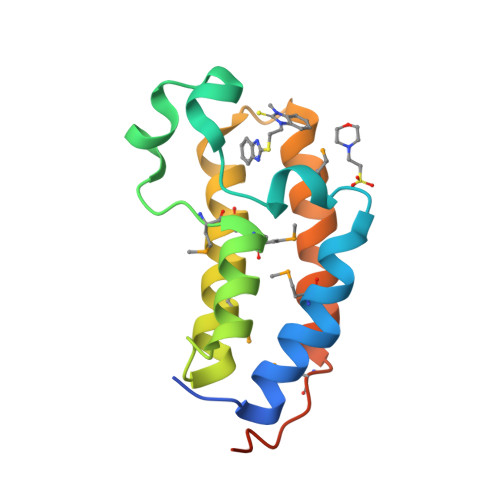
|
|
||||||||||||
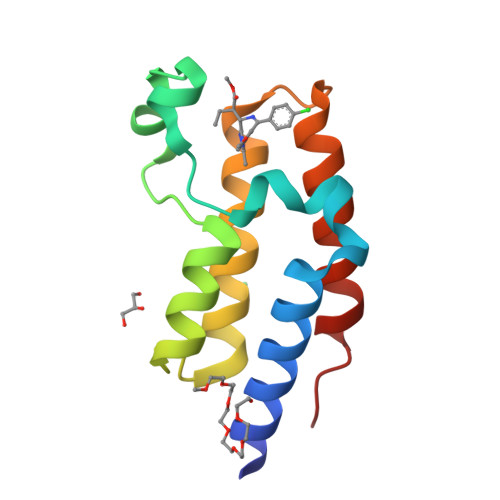
|
|
||||||||||||
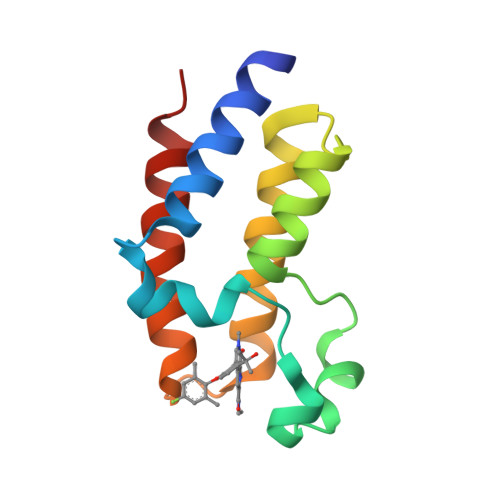
|
|
||||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antagonists | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Immuno Process Associations | |||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
| General Comments |
|
SARS-CoV-2/COVID-19 Experimental in vitro evidence, using affinity-purification mass spectrometry (AP-MS), indicates a protein-protein interaction between BRD2/4 and the SARS-CoV-2 envelope protein (E) [6], although whether this interaction is realistic based on spatial distribution of the host and viral proteins within cells was not addressed in this study. Speculatively, BRD inhibitors such as JQ1 and RVX 208, or BRD degraders (PROTACs; e.g. dBET6 or MZ1) could be utilised to examine the effect of inhibiting the BRD2/4-E interaction on SARS-CoV-2 pathobiology. |
1. Baud MGJ, Lin-Shiao E, Cardote T, Tallant C, Pschibul A, Chan KH, Zengerle M, Garcia JR, Kwan TT, Ferguson FM et al.. (2014) Chemical biology. A bump-and-hole approach to engineer controlled selectivity of BET bromodomain chemical probes. Science, 346 (6209): 638-641. [PMID:25323695]
2. Chung CW, Coste H, White JH, Mirguet O, Wilde J, Gosmini RL, Delves C, Magny SM, Woodward R, Hughes SA et al.. (2011) Discovery and characterization of small molecule inhibitors of the BET family bromodomains. J Med Chem, 54 (11): 3827-38. [PMID:21568322]
3. Dawson MA, Prinjha RK, Dittmann A, Giotopoulos G, Bantscheff M, Chan WI, Robson SC, Chung CW, Hopf C, Savitski MM et al.. (2011) Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature, 478 (7370): 529-33. [PMID:21964340]
4. Dittmann A, Werner T, Chung CW, Savitski MM, Fälth Savitski M, Grandi P, Hopf C, Lindon M, Neubauer G, Prinjha RK et al.. (2014) The commonly used PI3-kinase probe LY294002 is an inhibitor of BET bromodomains. ACS Chem Biol, 9 (2): 495-502. [PMID:24533473]
5. Faivre EJ, McDaniel KF, Albert DH, Mantena SR, Plotnik JP, Wilcox D, Zhang L, Bui MH, Sheppard GS, Wang L et al.. (2020) Selective inhibition of the BD2 bromodomain of BET proteins in prostate cancer. Nature, 578 (7794): 306-310. [PMID:31969702]
6. Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, O'Meara MJ, Rezelj VV, Guo JZ, Swaney DL et al.. (2020) A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature, 583 (7816): 459-468. [PMID:32353859]
7. Hewings DS, Wang M, Philpott M, Fedorov O, Uttarkar S, Filippakopoulos P, Picaud S, Vuppusetty C, Marsden B, Knapp S et al.. (2011) 3,5-dimethylisoxazoles act as acetyl-lysine-mimetic bromodomain ligands. J Med Chem, 54 (19): 6761-70. [PMID:21851057]
8. Imaide S, Riching KM, Makukhin N, Vetma V, Whitworth C, Hughes SJ, Trainor N, Mahan SD, Murphy N, Cowan AD et al.. (2021) Trivalent PROTACs enhance protein degradation via combined avidity and cooperativity. Nat Chem Biol, 17 (11): 1157-1167. [PMID:34675414]
9. Ito T, Umehara T, Sasaki K, Nakamura Y, Nishino N, Terada T, Shirouzu M, Padmanabhan B, Yokoyama S, Ito A et al.. (2011) Real-time imaging of histone H4K12-specific acetylation determines the modes of action of histone deacetylase and bromodomain inhibitors. Chem Biol, 18 (4): 495-507. [PMID:21513886]
10. Lucas SCC, Atkinson SJ, Chung CW, Davis R, Gordon L, Grandi P, Gray JJR, Grimes T, Phillipou A, Preston AG et al.. (2021) Optimization of a Series of 2,3-Dihydrobenzofurans as Highly Potent, Second Bromodomain (BD2)-Selective, Bromo and Extra-Terminal Domain (BET) Inhibitors. J Med Chem, 64 (15): 10711-10741. [PMID:34260229]
11. Lucas X, Wohlwend D, Hügle M, Schmidtkunz K, Gerhardt S, Schüle R, Jung M, Einsle O, Günther S. (2013) 4-Acyl pyrroles: mimicking acetylated lysines in histone code reading. Angew Chem Int Ed Engl, 52 (52): 14055-9. [PMID:24272870]
12. McLure KG, Gesner EM, Tsujikawa L, Kharenko OA, Attwell S, Campeau E, Wasiak S, Stein A, White A, Fontano E et al.. (2013) RVX-208, an inducer of ApoA-I in humans, is a BET bromodomain antagonist. PLoS ONE, 8 (12): e83190. [PMID:24391744]
13. Mirguet O, Lamotte Y, Chung CW, Bamborough P, Delannée D, Bouillot A, Gellibert F, Krysa G, Lewis A, Witherington J et al.. (2014) Naphthyridines as novel BET family bromodomain inhibitors. ChemMedChem, 9 (3): 580-9. [PMID:24000170]
14. Raina K, Lu J, Qian Y, Altieri M, Gordon D, Rossi AM, Wang J, Chen X, Dong H, Siu K et al.. (2016) PROTAC-induced BET protein degradation as a therapy for castration-resistant prostate cancer. Proc Natl Acad Sci USA, 113 (26): 7124-9. [PMID:27274052]
15. Seal J, Lamotte Y, Donche F, Bouillot A, Mirguet O, Gellibert F, Nicodeme E, Krysa G, Kirilovsky J, Beinke S et al.. (2012) Identification of a novel series of BET family bromodomain inhibitors: binding mode and profile of I-BET151 (GSK1210151A). Bioorg Med Chem Lett, 22 (8): 2968-72. [PMID:22437115]
16. Wang NY, Xu Y, Xiao KJ, Zuo WQ, Zhu YX, Hu R, Wang WL, Shi YJ, Yu LT, Liu ZH. (2020) Design, synthesis, and biological evaluation of 4,5-dihydro-[1,2,4]triazolo[4,3-f]pteridine derivatives as novel dual-PLK1/BRD4 inhibitors. Eur J Med Chem, 191: 112152. [PMID:32088495]
Bromodomain kinase (BRDK) family: bromodomain containing 2. Last modified on 23/08/2022. Accessed on 14/05/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetopharmacology.org/GRAC/ObjectDisplayForward?objectId=1944.