1. Ajit SK, Young KH. (2005) Analysis of chimeric RGS proteins in yeast for the functional evaluation of protein domains and their potential use in drug target validation.
Cell Signal, 17 (7): 817-25.
[PMID:15763424]
2. Almutairi F, Lee JK, Rada B. (2020) Regulator of G protein signaling 10: Structure, expression and functions in cellular physiology and diseases.
Cell Signal, 75: 109765.
[PMID:32882407]
3. Alqinyah M, Almutairi F, Wendimu MY, Hooks SB. (2018) RGS10 Regulates the Expression of Cyclooxygenase-2 and Tumor Necrosis Factor Alpha through a G Protein-Independent Mechanism.
Mol Pharmacol, 94 (4): 1103-1113.
[PMID:30049816]
4. Alqinyah M, Maganti N, Ali MW, Yadav R, Gao M, Cacan E, Weng HR, Greer SF, Hooks SB. (2017) Regulator of G Protein Signaling 10 (Rgs10) Expression Is Transcriptionally Silenced in Activated Microglia by Histone Deacetylase Activity.
Mol Pharmacol, 91 (3): 197-207.
[PMID:28031332]
5. Altman MK, Alshamrani AA, Jia W, Nguyen HT, Fambrough JM, Tran SK, Patel MB, Hoseinzadeh P, Beedle AM, Murph MM. (2015) Suppression of the GTPase-activating protein RGS10 increases Rheb-GTP and mTOR signaling in ovarian cancer cells.
Cancer Lett, 369 (1): 175-83.
[PMID:26319900]
6. Burgon PG, Lee WL, Nixon AB, Peralta EG, Casey PJ. (2001) Phosphorylation and nuclear translocation of a regulator of G protein signaling (RGS10).
J Biol Chem, 276 (35): 32828-34.
[PMID:11443111]
7. Cacan E, Ali MW, Boyd NH, Hooks SB, Greer SF. (2014) Inhibition of HDAC1 and DNMT1 modulate RGS10 expression and decrease ovarian cancer chemoresistance.
PLoS ONE, 9 (1): e87455.
[PMID:24475290]
8. DeHelian D, Gupta S, Wu J, Thorsheim C, Estevez B, Cooper M, Litts K, Lee-Sundlov MM, Hoffmeister KM, Poncz M et al.. (2020) RGS10 and RGS18 differentially limit platelet activation, promote platelet production, and prolong platelet survival.
Blood, 136 (15): 1773-1782.
[PMID:32542378]
9. Dhingra A, Faurobert E, Dascal N, Sterling P, Vardi N. (2004) A retinal-specific regulator of G-protein signaling interacts with Galpha(o) and accelerates an expressed metabotropic glutamate receptor 6 cascade.
J Neurosci, 24 (25): 5684-93.
[PMID:15215290]
10. Ewing RM, Chu P, Elisma F, Li H, Taylor P, Climie S, McBroom-Cerajewski L, Robinson MD, O'Connor L, Li M et al.. (2007) Large-scale mapping of human protein-protein interactions by mass spectrometry.
Mol Syst Biol, 3: 89.
[PMID:17353931]
11. Fang X, Chung J, Olsen E, Snider I, Earls RH, Jeon J, Park HJ, Lee JK. (2019) Depletion of regulator-of-G-protein signaling-10 in mice exaggerates high-fat diet-induced insulin resistance and inflammation, and this effect is mitigated by dietary green tea extract.
Nutr Res, 70: 50-59.
[PMID:30032988]
12. García-Bernal D, Dios-Esponera A, Sotillo-Mallo E, García-Verdugo R, Arellano-Sánchez N, Teixidó J. (2011) RGS10 restricts upregulation by chemokines of T cell adhesion mediated by α4β1 and αLβ2 integrins.
J Immunol, 187 (3): 1264-72.
[PMID:21705617]
13. Geurts M, Maloteaux JM, Hermans E. (2003) Altered expression of regulators of G-protein signaling (RGS) mRNAs in the striatum of rats undergoing dopamine depletion.
Biochem Pharmacol, 66 (7): 1163-70.
[PMID:14505795]
14. Gold SJ, Ni YG, Dohlman HG, Nestler EJ. (1997) Regulators of G-protein signaling (RGS) proteins: region-specific expression of nine subtypes in rat brain.
J Neurosci, 17 (20): 8024-37.
[PMID:9315921]
15. González-Castro TB, Martínez-Magaña JJ, Tovilla-Zárate CA, Juárez-Rojop IE, Sarmiento E, Genis-Mendoza AD, Nicolini H. (2019) Gene-level genome-wide association analysis of suicide attempt, a preliminary study in a psychiatric Mexican population.
Mol Genet Genomic Med, 7 (12): e983.
[PMID:31578828]
16. Haller C, Fillatreau S, Hoffmann R, Agenès F. (2002) Structure, chromosomal localization and expression of the mouse regulator of G-protein signaling10 gene (mRGS10).
Gene, 297 (1-2): 39-49.
[PMID:12384284]
17. Hensch NR, Karim ZA, Druey KM, Tansey MG, Khasawneh FT. (2016) RGS10 Negatively Regulates Platelet Activation and Thrombogenesis.
PLoS One, 11 (11): e0165984.
[PMID:27829061]
18. Hishimoto A, Shirakawa O, Nishiguchi N, Aoyama S, Ono H, Hashimoto T, Maeda K. (2004) Novel missense polymorphism in the regulator of G-protein signaling 10 gene: analysis of association with schizophrenia.
Psychiatry Clin Neurosci, 58 (5): 579-81.
[PMID:15482592]
19. Hooks SB, Callihan P, Altman MK, Hurst JH, Ali MW, Murph MM. (2010) Regulators of G-Protein signaling RGS10 and RGS17 regulate chemoresistance in ovarian cancer cells.
Mol Cancer, 9: 289.
[PMID:21044322]
20. Hooks SB, Murph MM. (2015) Cellular deficiency in the RGS10 protein facilitates chemoresistant ovarian cancer.
Future Med Chem, 7 (12): 1483-9.
[PMID:26293348]
21. Hooks SB, Waldo GL, Corbitt J, Bodor ET, Krumins AM, Harden TK. (2003) RGS6, RGS7, RGS9, and RGS11 stimulate GTPase activity of Gi family G-proteins with differential selectivity and maximal activity.
J Biol Chem, 278 (12): 10087-93.
[PMID:12531899]
22. Hunt TW, Fields TA, Casey PJ, Peralta EG. (1996) RGS10 is a selective activator of G alpha i GTPase activity.
Nature, 383 (6596): 175-7.
[PMID:8774883]
23. Kannarkat GT, Lee JK, Ramsey CP, Chung J, Chang J, Porter I, Oliver D, Shepherd K, Tansey MG. (2015) Age-related changes in regulator of G-protein signaling (RGS)-10 expression in peripheral and central immune cells may influence the risk for age-related degeneration.
Neurobiol Aging, 36 (5): 1982-93.
[PMID:25784210]
24. Kim W, Bennett EJ, Huttlin EL, Guo A, Li J, Possemato A, Sowa ME, Rad R, Rush J, Comb MJ et al.. (2011) Systematic and quantitative assessment of the ubiquitin-modified proteome.
Mol Cell, 44 (2): 325-40.
[PMID:21906983]
25. Kittanakom S, Barrios-Rodiles M, Petschnigg J, Arnoldo A, Wong V, Kotlyar M, Heisler LE, Jurisica I, Wrana JL, Nislow C et al.. (2014) CHIP-MYTH: a novel interactive proteomics method for the assessment of agonist-dependent interactions of the human β₂-adrenergic receptor.
Biochem Biophys Res Commun, 445 (4): 746-56.
[PMID:24561123]
26. Lee JK, Chung J, Druey KM, Tansey MG. (2012) RGS10 exerts a neuroprotective role through the PKA/c-AMP response-element (CREB) pathway in dopaminergic neuron-like cells.
J Neurochem, 122 (2): 333-43.
[PMID:22564151]
27. Lee JK, Chung J, Kannarkat GT, Tansey MG. (2013) Critical role of regulator G-protein signaling 10 (RGS10) in modulating macrophage M1/M2 activation.
PLoS ONE, 8 (11): e81785.
[PMID:24278459]
28. Lee JK, Chung J, McAlpine FE, Tansey MG. (2011) Regulator of G-protein signaling-10 negatively regulates NF-κB in microglia and neuroprotects dopaminergic neurons in hemiparkinsonian rats.
J Neurosci, 31 (33): 11879-88.
[PMID:21849548]
29. Lee JK, Kannarkat GT, Chung J, Joon Lee H, Graham KL, Tansey MG. (2016) RGS10 deficiency ameliorates the severity of disease in experimental autoimmune encephalomyelitis.
J Neuroinflammation, 13: 24.
[PMID:26831924]
30. Lee JK, McCoy MK, Harms AS, Ruhn KA, Gold SJ, Tansey MG. (2008) Regulator of G-protein signaling 10 promotes dopaminergic neuron survival via regulation of the microglial inflammatory response.
J Neurosci, 28 (34): 8517-28.
[PMID:18716210]
31. Lu J, Gosslau A, Liu AY, Chen KY. (2008) PCR differential display-based identification of regulator of G protein signaling 10 as the target gene in human colon cancer cells induced by black tea polyphenol theaflavin monogallate.
Eur J Pharmacol, 601 (1-3): 66-72.
[PMID:18992738]
32. Luck K, Kim DK, Lambourne L, Spirohn K, Begg BE, Bian W, Brignall R, Cafarelli T, Campos-Laborie FJ, Charloteaux B et al.. (2020) A reference map of the human binary protein interactome.
Nature, 580 (7803): 402-408.
[PMID:32296183]
33. Ma P, Cierniewska A, Signarvic R, Cieslak M, Kong H, Sinnamon AJ, Neubig RR, Newman DK, Stalker TJ, Brass LF. (2012) A newly identified complex of spinophilin and the tyrosine phosphatase, SHP-1, modulates platelet activation by regulating G protein-dependent signaling.
Blood, 119 (8): 1935-45.
[PMID:22210881]
34. Ma P, Foote DC, Sinnamon AJ, Brass LF. (2015) Dissociation of SHP-1 from spinophilin during platelet activation exposes an inhibitory binding site for protein phosphatase-1 (PP1).
PLoS ONE, 10 (3): e0119496.
[PMID:25785436]
35. Ma P, Gupta S, Sampietro S, DeHelian D, Tutwiler V, Tang A, Stalker TJ, Brass LF. (2018) RGS10 shapes the hemostatic response to injury through its differential effects on intracellular signaling by platelet agonists.
Blood Adv, 2 (16): 2145-2155.
[PMID:30150297]
36. Mao Y, Lei L, Su J, Yu Y, Liu Z, Huo Y. (2014) Regulators of G protein signaling are up-regulated in aspirin-resistant platelets from patients with metabolic syndrome.
Pharmazie, 69 (5): 371-3.
[PMID:24855830]
37. Miao R, Lu Y, Xing X, Li Y, Huang Z, Zhong H, Huang Y, Chen AF, Tang X, Li H et al.. (2016) Regulator of G-Protein Signaling 10 Negatively Regulates Cardiac Remodeling by Blocking Mitogen-Activated Protein Kinase-Extracellular Signal-Regulated Protein Kinase 1/2 Signaling.
Hypertension, 67 (1): 86-98.
[PMID:26573707]
38. Oláh J, Vincze O, Virók D, Simon D, Bozsó Z, Tõkési N, Horváth I, Hlavanda E, Kovács J, Magyar A et al.. (2011) Interactions of pathological hallmark proteins: tubulin polymerization promoting protein/p25, beta-amyloid, and alpha-synuclein.
J Biol Chem, 286 (39): 34088-100.
[PMID:21832049]
39. Riekenberg S, Farhat K, Debarry J, Heine H, Jung G, Wiesmüller KH, Ulmer AJ. (2009) Regulators of G-protein signalling are modulated by bacterial lipopeptides and lipopolysaccharide.
FEBS J, 276 (3): 649-59.
[PMID:19120454]
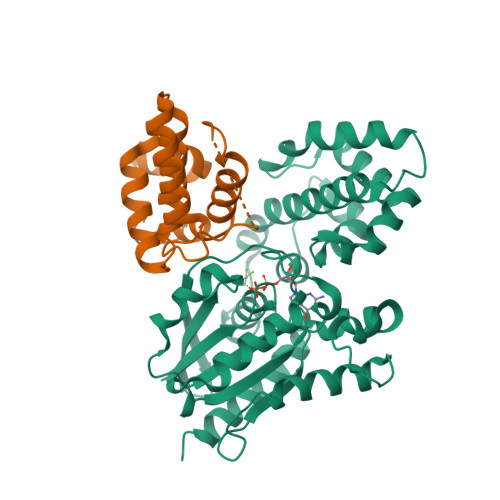
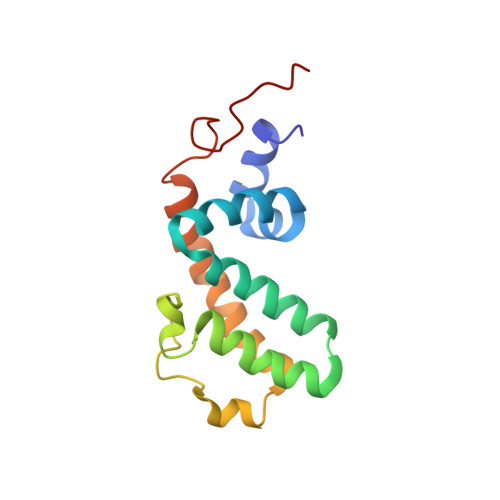
40. Soundararajan M, Willard FS, Kimple AJ, Turnbull AP, Ball LJ, Schoch GA, Gileadi C, Fedorov OY, Dowler EF, Higman VA et al.. (2008) Structural diversity in the RGS domain and its interaction with heterotrimeric G protein alpha-subunits.
Proc Natl Acad Sci USA, 105 (17): 6457-62.
[PMID:18434541]
41. Stes E, Laga M, Walton A, Samyn N, Timmerman E, De Smet I, Goormachtig S, Gevaert K. (2014) A COFRADIC protocol to study protein ubiquitination.
J Proteome Res, 13 (6): 3107-13.
[PMID:24816145]
42. Sun H, Calipari ES, Beveridge TJ, Jones SR, Chen R. (2015) The brain gene expression profile of dopamine D2/D3 receptors and associated signaling proteins following amphetamine self-administration.
Neuroscience, 307: 253-61.
[PMID:26321241]
43. Talele S, Gonzalez S, Trudeau J, Junaid A, Loy CA, Altman RA, Sjögren B. (2024) A Phenotypic High-Throughput Screen Identifies Small Molecule Modulators of Endogenous RGS10 in BV-2 Cells.
J Med Chem, 67 (22): 20343-20352.
[PMID:39547663]
44. Tu Y, Popov S, Slaughter C, Ross EM. (1999) Palmitoylation of a conserved cysteine in the regulator of G protein signaling (RGS) domain modulates the GTPase-activating activity of RGS4 and RGS10.
J Biol Chem, 274 (53): 38260-7.
[PMID:10608901]
45. Wagner SA, Beli P, Weinert BT, Nielsen ML, Cox J, Mann M, Choudhary C. (2011) A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles.
Mol Cell Proteomics, 10 (10): M111.013284.
[PMID:21890473]
46. Waugh JL, Lou AC, Eisch AJ, Monteggia LM, Muly EC, Gold SJ. (2005) Regional, cellular, and subcellular localization of RGS10 in rodent brain.
J Comp Neurol, 481 (3): 299-313.
[PMID:15593368]
47. Yang S, Chen W, Stashenko P, Li YP. (2007) Specificity of RGS10A as a key component in the RANKL signaling mechanism for osteoclast differentiation.
J Cell Sci, 120 (Pt 19): 3362-71.
[PMID:17881498]
48. Yang S, Hao L, McConnell M, Zhou X, Wang M, Zhang Y, Mountz JD, Reddy M, Eleazer PD, Li YP et al.. (2013) Inhibition of Rgs10 Expression Prevents Immune Cell Infiltration in Bacteria-induced Inflammatory Lesions and Osteoclast-mediated Bone Destruction.
Bone Res, 1 (3): 267-281.
[PMID:24761229]
49. Yang S, Li YP. (2007) RGS10-null mutation impairs osteoclast differentiation resulting from the loss of [Ca2+]i oscillation regulation.
Genes Dev, 21 (14): 1803-16.
[PMID:17626792]
50. Zhang Y, Zhang C, Li H, Hou J. (2019) The Presence of High Levels of Circulating Trimethylamine N-Oxide Exacerbates Central and Peripheral Inflammation and Inflammatory Hyperalgesia in Rats Following Carrageenan Injection.
Inflammation, 42 (6): 2257-2266.
[PMID:31489527]


 target has curated data in GtoImmuPdb
target has curated data in GtoImmuPdb